- Your cart is empty
- Continue Shopping
iMatrix-511 Silk Recombinant Laminin E8 Fragments
iMatrix-511 stands out as an advanced extracellular matrix compatible with a diverse range of cell types. Particularly ideal for pluripotent and mesenchymal stem cells, this product features recombinant Laminin-511 E8 protein fragments. These fragments facilitate the maintenance of ES/iPS cells and MSCs in xeno-free culture environments. Moreover, iMatrix-511 supports the single-cell passaging process and offers superior adhesion compared to full-length Laminin, Vitronectin, or Matrigel.
iMatrix-511 Silk is purified from silkworm in which gene of human Laminin-511 E8 fragment is incorporated. iMatrix-511 Silk has the same functions and performance as iMatrix-511 at a more competitive pricing.
Key Features:
- Ideal for Xeno-Free, Feeder-Free Cell Culture
- Proven to provide superior adhesion of human ES and iPS cells
- Enables the passaging of single cells
- Compatible with a wide variety of cell types
- Facilitates the extended culture of hES/hiPS cells and hMSCs
- No pre-coating required
Clinical Grade iMatrix-511:
The clinical grade version of iMatrix-511, iMatrix-511MG, meets the Standards of Biological Ingredients by Pharmaceutical and Medical Devices Agency (PMDA), a government agency in Japanese that is homologous to the FDA. The regulation is one of the strictest in the world and iMatrix-511MG not only fulfils regulatory requirements, it has since been used for a number of high-profile clinical applications in Japan – Center for iPS Cell Research & Application (CiRA) Clinical iPSC Stock Project and Kyoto University Institute for Frontier Life & Medical Sciences Embryonic Stem Cell Banking Project. iMatrix-511 MG was used in the world’s first successful iPSC corneal transplantation at Osaka University.
Checkout the links for more information:
- World’s first transplant of cornea made from iPS cells | Graduate School of Medicine / Faculty of Medicine, Osaka University (osaka-u.ac.jp)
- Co-ordinated ocular development from human iPS cells and recovery of corneal function – PubMed (nih.gov)
- Coordinated generation of multiple ocular-like cell lineages and fabrication of functional corneal epithelial cell sheets from human iPS cells – PubMed (nih.gov)
- Overview: an iPS cell stock at CiRA – PubMed (nih.gov)
Matrix-511MG is not only GMP-compatible but also GMP-adaptable, making it a versatile resource. Transition seamlessly from basic research, utilizing our RUO iMatrix-511, to clinical applications with iMatrix-511MG. Our clinical-grade products offer the same versatility and benefits as our RUO products. Benefit from time and cost savings with the Pre-Mix Method, along with increased detachment efficiency and cell viability through the Scraper-Free Method.
Check out iMatrix from this youtube link:
Technical Information
Laminin-511 E8 fragment
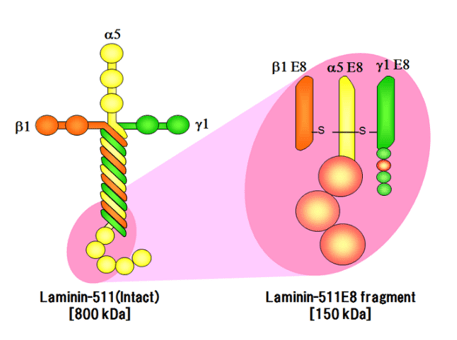
Laminin-511, a significant constituent of the basement membrane, serves as a scaffold for pluripotent stem cells such as ES/iPS cells. As part of the basement membrane, laminin-511 (800 kDa) interacts with integrins on cell surfaces. Comprising three chains (α-, β-, and γ-chains) and forming supramolecular aggregates, laminin-511 is a notably large protein (800 kDa) that poses challenges for recombinant production. The Laminin-511 E8 fragment (150 kDa) has been identified as the smallest component capable of binding to integrins on the cell surface.
Greater Cell Adhesion & Proliferation
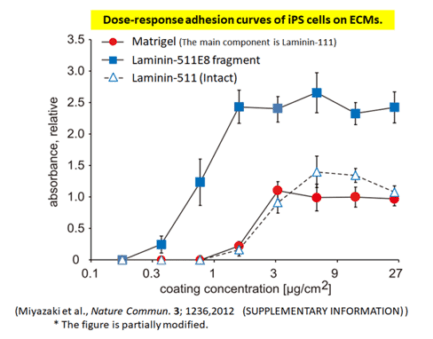
Human iPS cells adhere more strongly to Laminin-511 E8 fragment than to the Laminin-511 (Intact) or Matrigel.
Maintain and Culture Single ES/iPS Cells with Greater Propagation
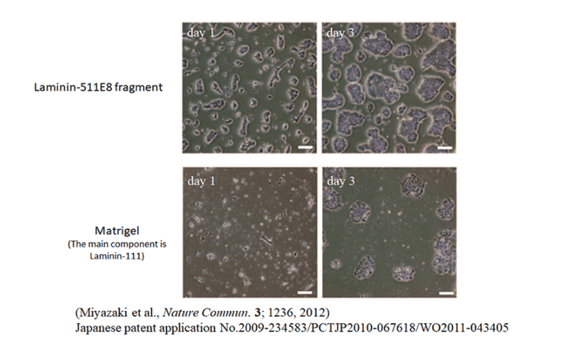
Dispersed human single ES cells adhere rapidly to Laminin-511 E8 fragment and proliferate immediately.
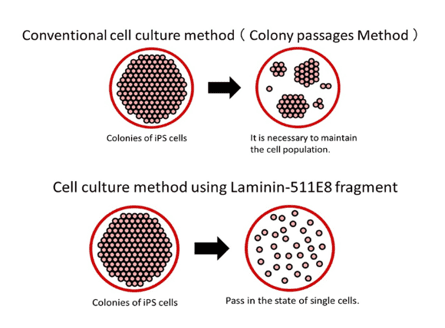
Conventional culture methods often lead to cell death when ES/iPS cells are dispersed into single cells. Consequently, maintaining a cell colony becomes essential during the passaging process. However, utilizing the Laminin-511 E8 fragment as a cell culture substrate enables the successful maintenance and cultivation of even a single ES/iPS cell.
Enhance Growth of ES/iPS Cell Culture
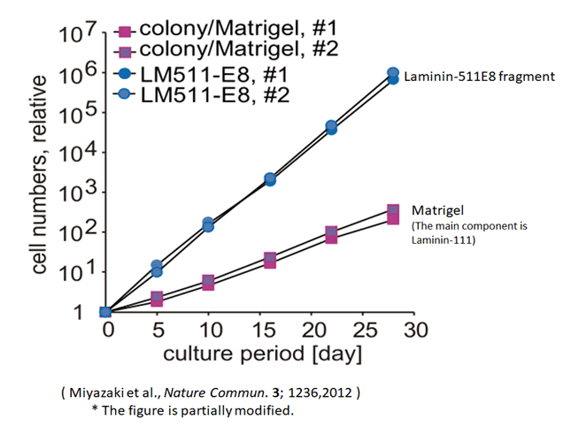
The use of Laminin-511 E8 fragment increases the rate of proliferation as compared to the use of Matrigel.
No Pre-Cating Required
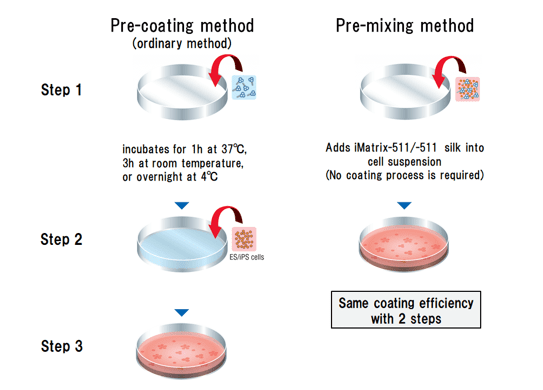
The pre-mixed method is both time-saving and cost-effective as it only requires half the amount of substrate when compared to the pre-coating method.
Reference Publications
Makoto Ryosaka, Shin-Ichi Mae, Kenji Osafune
Protocol for the generation and expansion of human iPS cell-derived ureteric bud organoids
STAR Protocols 3, 101484
DOI: https://doi.org/10.1016/j.xpro.2022.101484
Article Snippet: “We add iMatrix-511 silk to the medium to coat the plate at passaging. You may precoat the plate with iMatrix-511 silk at 37°C for 1 h; however, this is a time-consuming process”
Ishikawa, Mitsuru et al.
miRNA-Based Rapid Differentiation of Purified Neurons from hPSCs Advancestowards Quick Screening for Neuronal Disease Phenotypes In Vitro
Cells vol. 9,3 532. 25 Feb. 2020
DOI: 10.3390/cells9030532
Article Snippet: “seeded at 1.5 × 105 cells/well of tissue culture treated six-well plate in 1.5 or 2.0 mL medium with 1.5 µg/ml iMatrix-511 silk (Laminin-511 E8, Nippi, Tokyo, Japan) in the presence of 10 µM”
Shin-Ichi Mae, Makoto Ryosaka, Satoko Sakamoto, et al.
Expansion of Human iPSC-Derived Ureteric Bud Organoids with Repeated Branching Potential
Cell Reports 32, 107963,
DOI: https://doi.org/10.1016/j.celrep.2020.107963
Article Snippet: “After lipofection, the cells were plated onto one well of a 6-well iMatrix-511 silk-coated plate in StemFit AK02N medium with 10 mM Y-27632.”
Hirayama, R., Toyohara, K., Watanabe, K. et al.
iPSC-derived type IV collagen α5-expressing kidney organoids model Alport syndrome
Commun Biol 6, 854 (2023).
DOI: https://doi.org/10.1038/s42003-023-05203-4
Article Snippet: “iPSCs cultured in StemFit® AK02N and iMatrix-511 silk were differentiated into NPCs using DMEM/F12 GlutaMAX.”
Mio Iwasaki, Nicola J. Drummond, David E. McNay et al.
Non-invasive monitoring of midbrain dopaminergic progenitor cell production from human pluripotent stem cells
01 September 2023, PREPRINT (Version 1) available at Research Square
DOI: https://doi.org/10.21203/rs.3.rs-2996413/v1
Article Snippet: “At day 11, the cells were lifted with Accumax (Innovative Cell Technologies) and replated to iMatrix-511 silk-coated 24-well plates at a density of 500,000 cells/cm2.”
Chen, J., Horiuchi, S., Kuramochi, S. et al.
Human intestinal organoid-derived PDGFRα + mesenchymal stroma enables proliferation and maintenance of LGR4 + epithelial stem cells
Stem Cell Res Ther 15, 16 (2024).
DOI: https://doi.org/10.1186/s13287-023-03629-5
Article Snippet: “Intestinal organoids were cut and spread on a 35-mm dish coated with iMatrix-511 silk. The dishes were coated with 1.7 μg/ml iMatrix-511 silk at 37 °C for 1 h.”
Hiranuma, M., Okuda, Y., Fujii, Y. et al.
Characterization of human iPSC-derived sensory neurons and their functional assessment using multi electrode array
Sci Rep 14, 6011 (2024).
DOI: https://doi.org/10.1038/s41598-024-55602-8
Article Snippet: “The conical tube was centrifuged at 300 × g for 5 min at room temperature. When using a 24 well plate coated with iMatrix-511 silk (Nippi), the sensory neurons were seeded at 1.5 × 105 cells per well at a density of 3 × 105 cells/mL.”
Kazutoshi Takahashi, Chikako Okubo, Michiko Nakamura, et al.
A stress-reduced passaging technique improves the viability of human pluripotent cells
Cell Reports Methods 2, 100155
DOI: https://doi.org/10.1016/j.crmeth.2021.100155
Article Snippet: Highlight from the Chemicals, peptides, media and recombinant proteins table “iMatrix-511 silk Nippi Cat# 892021″
Documents
FAQs
1. What format(s) are the iMatrixTM products sold in?
- Currently, it is sold only in liquid form. One tube contains 175 ug of cell culture substrate (laminin 511-E8 fragment) at a concentration of 0.5 mg / mL. * The lyophilized version of iMatrixTM-511 has been discontinued by the manufacturer as of March 2015.
2. What is the product storage temperature and expiration date?
- The storage temperature of the product is refrigerated at 2-8 ° C. Please refer to the table below for product expiration date.
| Expiration date | Applicable products |
| 2 years from date of production | iMatrixTM-511, iMatrixTM-511 silk, iMatrixTM-511MG, iMatrixTM-411, iMatrixTM-221 |
[ The specific expiration date is printed on the product box. ]
3. Is it possible to store iMatrixTM frozen? (-80 °C)
- No. It can not be stored frozen.
4. What is the purity of the iMatrixTM products?
- Purity is over 95%. Please refer to the lot specific data sheet for more information.
5. What is the recommended medium for culturing hES/hiPS cells when using iMatrixTM products?
- Miyazaki et al. Nature communications, 3(1236), 1-10, 2012, and Scientific Reports, 7, 41165, 2017. The following are used. ・mTeSR1,TeSR2,TeSR-E8 (STEMCELL Technologies) ・StemProhESCSFM (Thermo Fisher Scientific) ・StemFitAK03 (Ajinomoto) Nakagawa et al. Scientific Reports, 4(3594), 1-7, 2014. The following are used. ・StemFit (Ajinomoto) The publication stated that all media showed good results.
6. What is the optimal coating concentration for iPS cell culture?
- The optimal coating concentration depends on the cell line and must be determined by the end user. We recommend first trying iMatrixTM at 0.5 ug / cm2 and consider the optimal concentration to be between 0.1 and 1.5 ug / cm2. In addition, there is also a new method for plating, the Pre-mix method: Miyazaki et al. Scientific Reports, 7, 41165, 2017.
7. Is there a culture protocol for iPS cells using iMatrixTM-511?
- Please refer to the link below for the expansion culture protocol of ES / iPS cells using iMatrixTM-511((Expansion culture protocol))
8. Is Rock Inhibitor (Y-27632) necessary for culturing iPS cells?
- Nakagawa et al. Scientific Reports, 4(3594), 2014. Use Rock Inhibitor at cell passage. However, it is not used during medium exchange.
9. Can iPS cell passage be handled with a single cell?
- Yes. iPS cell passage can be handled with a single cell. Nakagawa et al. Scientific Reports, 4(3594), 2014. * Links to the external site. ((Expansion culture protocol))
10. What is used to detached the cells?
- We recommend using trypsin. Nakagawa et al. Scientific Reports, 4(3594), 2014. * Links to the external site. ((Expansion culture protocol))
11. Can this product be used for mouse iPS cells?
- Currently, there is no data to support culture of mouse iPS cells using iMatrixTM-511.
12. What is the difference between iMatrixTM-511 and Matrigel?
- Matrigel contains laminin-111 from mouse EHS sarcoma. It also contains molecules other than laminin. iMatrixTM-511 is a recombinant protein purified from laminin 511-E8 fragment expressed in CHO-S cells with high purity. iMatrixTM-511 silk is a recombinant protein in which the laminin 511-E8 fragment is highly purified by silkworm-produced cocoons. Human ES cells and iPS cells are known to adhere to laminin-511 through cell membrane receptors (especially a6b1 integrin). Human ES cells and iPS cells are known to exhibit high adhesion activity to laminin-511. This makes it possible to pass iPS cells into single cells and pass them with iMatrixTM-511 / iMatrixTM-511 silk. In Miyazaki et al. (Nature communications, 3 (1236), 1-10, 2012), the expansion efficiency of the number of cells after 5 passages (after 30 days) is about 200 times higher than that of Matrigel.