- Your cart is empty
- Continue Shopping
Quality Control Testing Services GemPharmatech
GemPharmatech has achieved AAALAC and IS09001 quality system certification and has built a state-of-the-art animal husbandry management and health monitoring system to ensure the animals’ care, health, and quality. We are committed to providing our customers with consistently high-quality animals and services. We’ve also established more than 10,000 genetically modified mouse models, and can provided consulting services on various model products. Our team values communication and interaction with our clients.
ANIMAL QUALITY TESTING SERVICES
SNP Testing Services
SNP Testing Services Overview
Single nucleotide polymorphism (SNP) testing is used for annual monitoring of the genetic background of rats and mice, detection of gene mutations or genetic contaminations and batch testing of rat and mouse mutants.
Why GemPharmatech
One-stop solution: We provide a one-stop solution for SNP testing from primer design to result reporting.
Rich experience: Since establishing the high-throughput testing platform, we have cumulatively tested more than 100,000 data points.
Stable genetic testing panel: We have successfully developed SNP panels applicable to genetic testing and speed congenics for 13 inbred mouse strains.
SNP Testing Services
SNP testing refers to the conversion, transversion, insertion, and deletion of a single base arising from the mutation of a single nucleotide with a mutation frequency greater than 1% in a population. As a third-generation genetic molecular marker, the SNP labeling technique possesses numerous advantages in genetic quality monitoring as compared with other genetic markers, including wide distribution, good genetic stability, impressive representativeness, and easier implementation of high-throughput and automatic monitoring. As a result, it has been widely applied in agriculture, pharmaceutical, life science, and other research fields.
We can perform SNP testing using KASP (Kompetitive Allele Specific PCR) for the following purposes:
- Annual monitoring of genetic background of rats and mice: Annual genetic testing for laboratory animals is an essential approach to evaluate and ensure the quality of laboratory animals, especially for which regular genetic background monitoring is necessary.
- Testing for genetic mutation and contamination in rats and mice: This test should be performed when a new phenotype is identified or when a mismatched rat/mouse is suspected during a study.
- Batch testing of rat and mouse mutants: SNP testing can be used for precise genotyping of InDels (insertions and deletions).
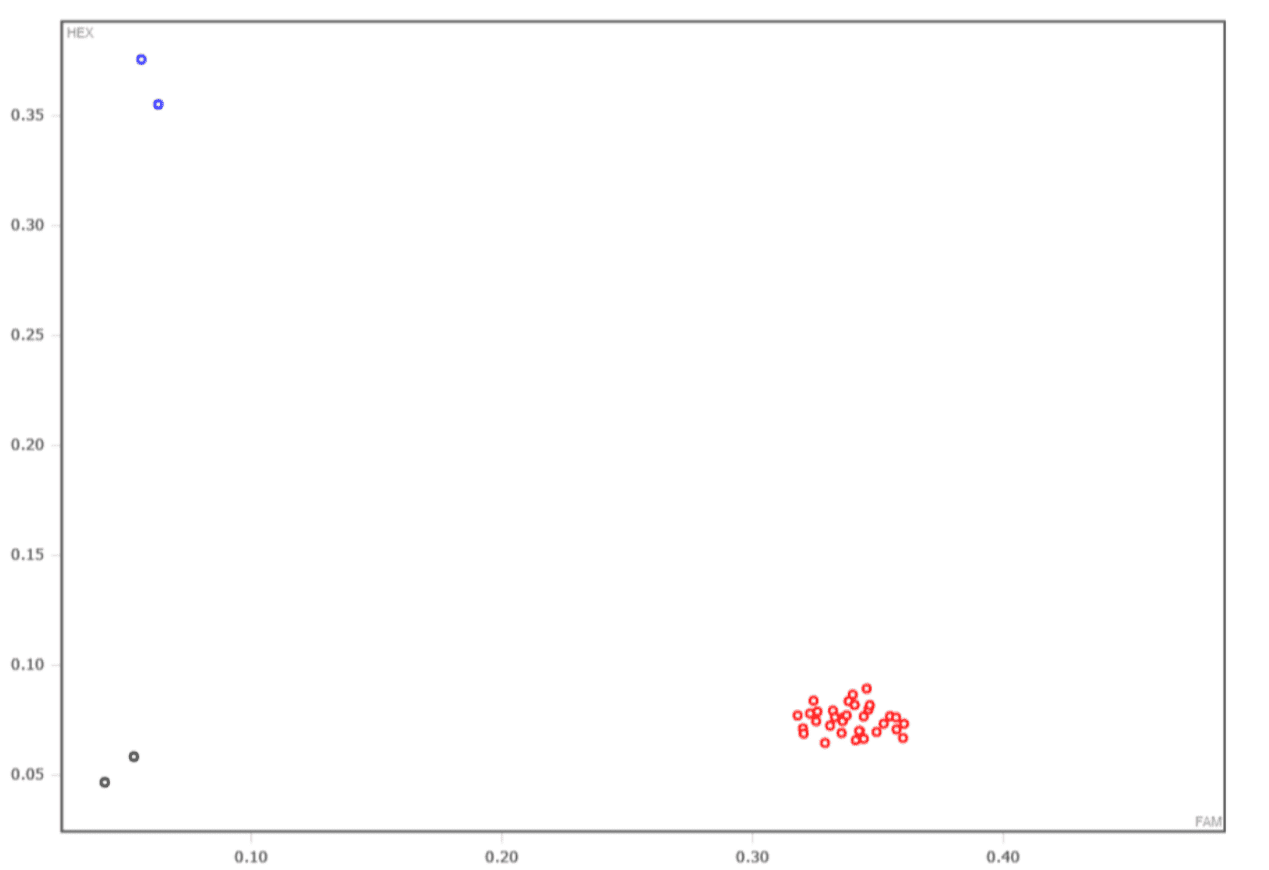
X: HEX Channel Y: FAM Channel
Red dots:FAM/FAM, Blue dots: HEX/HEX, Black dots: blank control. - High-throughput KASP based on available SNP loci information is utilized for precise allele genotyping of specified SNPs and InDels in diverse genome samples (including complicated genomic DNA samples), employing fluorescence detection with high sensitivity. In terms of technological principle, it is identical to Taqman assay (the gold standard) and produces consistent accuracy. Unlike traditional Taqman assays, however, amplification of the general fluorescence primer is available for assay at all loci based on the unique ARM PCR approach, rather than requiring a probe or primer generated for each target. We have optimized PCR methods to address the needs of high-throughput reactions at various loci. While the accuracy of KASP is comparable to the gold standard Taqman test, it is more economical and has better loci adaptability.
Microbiology Testing Services
Microbiology Testing Overview
Hygienic, standardized SPF-grade/germ-free mice are the foundation of successful research. GemPharmatech is more than just a top tier mouse provider. Our professional microbiology testing center can also execute precise and high-quality microbiology tests on SPF-grade mice, germ-free mice, and environment-related microorganisms. GemPharmatech is committed to providing quality resources for your research by focusing on the health of laboratory animals.
The microbiology testing platform team is highly qualified with 10+ team members and six testing laboratories. The testing center is equipped with a VITEK2 automatic bacterial identification instrument; triple membrane filtration system; mold, fungi, bacteria, anaerobic incubators; and other testing devices, which can be used to detect the full range of bacteria, viruses, and parasites for mice and related environmental samples. We have passed numerous certification proficiency tests conducted by local and international laboratories for many years.
Why GemPharmatech
Capability: GemPharmatech passed the ICLAS (International Council for Laboratory Animal Science) testing capability evaluation, testing level, and international requirements.
Reliability: Diversified detection approaches. For focused detection, several methods can be applied to various sample types.
High throughput: Various testing methods and automated equipment considerably improve detection efficiency and sample throughput.
Microbiology testing services
- Viral testing
- Bacterial testing
- Parasitic testing
- Sterility testing
1. Pathogenic and microorganisms tests of mice
| Agents |
Essential test items for SPF-grade animals specified by national standards |
All test items for SPF- grade animals specified by national standards |
Methods |
|||
| Microscopy | Culture | ELISA | PCR(qPCR) | |||
| VIRUS | ||||||
| Ectromelia Virus (ECT) | ● | ● | ✔ | ✔ | ||
| Mouse hepatitis virus (MHV) | ● | ● | ✔ | ✔ | ||
| Sendai virus (SV) | ● | ● | ✔ | ✔ | ||
| Pneumonia virus of Mouse (PVM) | ● | ● | ✔ | ✔ | ||
| Reovirus type 3 (Reo-3) | ● | ● | ✔ | ✔ | ||
| Minute virus of Mouse (MVM) | ● | ● | ✔ | ✔ | ||
| Lymphocytic choriomeningitis virus (LCMV) | ● | ✔ | ✔ | |||
| Hanta virus (HV) | ● | ✔ | ✔ | |||
| Theiler’s mouse encephalomyelitis virus (TMEV) | ● | ✔ | ✔ | |||
| Mouse adenovirus type 1 (FL) & 2(K87) | ● | ✔ | ✔ | |||
| Polyoma virus (Poly) | ● | ✔ | ✔ | |||
| Mouse Norovirus (MNV) | ✔ | ✔ | ||||
| Mouse cytomegalovirus (MCMV) | ✔ | |||||
| Epizootic Diarrhea of Infant Mice (EDIM) | ✔ | |||||
| K virus (K) | ✔ | ✔ | ||||
| Mouse Parvovirus (MPV) | ✔ | ✔ | ||||
| Lactate dehydrogenaseelevating virus(LDV) | ✔ | |||||
| Mouse kidney parvovirus(MKPV) | ✔ | |||||
| BACTERIA | ||||||
| Salmonella spp. | ● | ● | ✔ | ✔ | ||
| Corynebacterium kutscheri | ● | ● | ✔ | ✔ | ||
| Pasteurella pneumotropica | ● | ● | ✔ | ✔ | ||
| Klebsiella pneumoniae | ● | ● | ✔ | ✔ | ||
| Staphylococcus aureus | ● | ● | ✔ | ✔ | ||
| Pseudomonas aeruginosa | ● | ● | ✔ | ✔ | ||
| Streptobacillus moniliformis | ● | ✔ | ✔ | |||
| Streptococcus pneumoniae | ● | ✔ | ✔ | |||
| β-hemolyticstreptococcus | ● | ✔ | ✔ | |||
| Escherichia coli O115 a,C,K(B) | ● | ✔ | ✔ | |||
| Klebsiella oxytoca | ✔ | ✔ | ||||
| Citrobacter rodentium | ✔ | ✔ | ||||
| Proteus mirabils | ✔ | ✔ | ||||
| Pasteurella multocida | ✔ | ✔ | ||||
| Corynebacterium bovis | ✔ | ✔ | ||||
| Mycoplasma spp. | ● | ● | ✔ | ✔ | ||
| Clostridium piliforme (Tyzzer’s) | ● | ● | ✔ | ✔ | ||
| Cilia-associate respiratory bacillus | ✔ | ✔ | ||||
| Yersinia enterocolitica | ● | ✔ | ||||
| Yersinia pseudotuberculosis | ● | ✔ | ||||
| Pathogenic dermal fungi | ● | ✔ | ||||
| Helicobacter spp. | ✔ | |||||
| PARASITES | ||||||
| Ectoparasites | ● | ● | ✔ | |||
| All Helminths | ● | ● | ✔ | |||
| Flagellates | ● | ● | ||||
| Ciliates | ● | ● | ||||
| Toxoplasma gondii | ● | ● | ✔ | |||
| Encephalitozoon cuniculi | ● | ✔ | ||||
| Pneumocystis carinii | ● | ✔ | ||||
| Entamoeba | ||||||
2. Sterility test
| Testing Items | Methods |
|---|---|
| Germ-free mouse testing | Isolation culture |
| Feed sterility testing | Isolation culture |
| Bedding sterility testing | Isolation culture |
| Drinking water sterility testing | Isolation culture |
| Environmental swab for sterility testing | Isolation culture |
| Colony testing | Isolation culture |
Genetic Integrity
Breeding System
In recent years, as the biomedical industry has flourished, the resources of laboratory rodent strains have expanded, and the demand for quality laboratory rodents has increased. A critical concern for laboratory animal providers is to avoid genetic drifting and genetic contamination in order to deliver high-quality and reliable laboratory animals to researchers.
GemPharmatech has developed a strict breeding management system that ensures the genetic quality of various mouse strains is consistent with international standards and maintains genetic stability in long-term breeding and commercial production. This system was developed based on our many years of experience in breeding laboratory animals.
The central premise of breeding all inbred and pure strains of mice is that the animals’ homozygosity and genetic purity must be maintained, and all strains must have detailed records of strain breeding and pedigree charts. Our breeding management system strictly controls and reduces genetic contamination and drift during the breeding process, providing researchers with high-quality animals that have consistent and genetically stable features.
Our mouse population structure is a compact pyramid, with production lines consisting of a foundation group, a pedigree expansion group, and a production group.
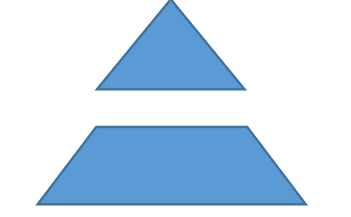 |
Foundation group: sibling pairs, up to three generations aremaintained simultaneously; cryopreservation of genetic material and resuscitation after 10 generations of continuous breeding;
annual SNP monitoring. Pedigree expansion group: all sibling pairings are within 2 generations, up to 5 generations of common ancestors, annual SNP monitoring with statistical assessment. |
 |
Product group: all sibling pairings are within 2 generations, up to 5-10 generations of common ancestors, annual SNP monitoring with statistical assessment. |
We ensure continuous and uninterrupted breeding of a large number of mouse strains for at least 50 years by employing this system. The population has only passed down two generations, reducing the risk of genetic variation and drift during breeding.
Genetic Monitoring Systems
GemPharmatech uses single nucleotide polymorphisms (SNPs) to track the genetics of live breeding colonies at each of our facilities.
GemPharmatech chose 19 autosomes and 1 X chromosome for various inbred strains, selected monitoring loci on each chromosome at approximately equal distances, and targeted some strain-specific genetic loci by consulting academic papers to construct a library of nearly one thousand specific SNP monitoring loci. On this premise, we have built a panel of 96 SNPs for each inbred or isogenic inbred line, which is adequate to distinguish all the in-house inbred strains and to ensure the genetic stability of each line.
GemPharmatech conducts annual genetic monitoring of the foundation groups of select lines and the production groups of select lines. In addition, we sample inbred background lines each year to ensure the stable genetic background for all newly engineered mouse strains.
Animal Health
GemPharmatech has achieved AAALAC and ISO 9001 quality system certification and has built a state-of-the-art animal husbandry management and health monitoring system to ensure the animals’ care, health, and quality. GemPharmatech is committed to providing our customers with consistently high-quality animals and services.
Staff management
- All staff entering animal facilities must receive an animal experiment introduction certificate, undergo rigorous training before entering the facilities, and only gain access to animal facilities after completing the training.
- All staff must enter with access cards, adhere strictly to the personnel entry and exit procedure, wear disposable protective clothing (shoes, gloves, hair caps, masks, and a complete set of PPE (disposable protective clothing, shoes, gloves, hair caps and masks), and shower before entering the production area.
- To reduce the risk of cross-contamination, all staff members must restrict the use of facilities and rooms.
Breeding Management
Breeding Facilities
The GemPharmatech facility is a barrier environment, and SPF mice are housed in centrally ventilated IVC cages and proprietary IVU cages.

Feed
Co60 irradiated and sterilized vacuum-packed pellet feed is obtained from qualified suppliers, and third-party test reports are provided annually, including testing for nutrient content, heavy metals, pesticide residues, and microbes.
The storage warehouse is controlled for temperature, humidity, and pests. The feed is positioned off the wall and the floor and each batch of feed is sampled and tested for sterility before being used in the facility. Mice are fed ad libitum. Feeding containers are replaced every one to two weeks.
Bedding and nesting materials
Corn cob bedding and shavings bedding are procured from qualified suppliers, and third-party test reports are included. Testing on hygiene, chemical contamination, and microbes are conducted annually. Each batch of bedding material is inspected for particle size, color, odor, and contaminants and is stored away from walls and floors. The bedding material is autoclaved before entering the facility.
Drinking water
In order to provide mice with clean water, GemPharmatech utilizes the Edstrom Automatic Drinking Water System with RO reverse osmosis and chlorine treatment. The drinking water is routinely tested for sterility.
Disinfection
The GemPharmatech facility is equipped with several sets of advanced production support equipment. We have high-capacity sterilizing equipment, a multi-floor hydrogen peroxide fumigation and disinfection system, and an automated production line for centralized bedding processing, cleaning, and refilling. Before entering the facility, all materials are sterilized, and all rooms and items are disinfected.

Animal health monitoring
GemPharmatech employs a monitoring system to assure the microbiological quality , decontaminated offspring, environmental (dust) monitoring of breeding mice. The test samples include sentinel mice and breeding stock mice. The test consists of 48 items. In addition, animals are sampled quarterly and sent to third-party testing institutions for monitoring.
Each side of the rack contains a sentinel mouse cage to determine the microbiological status of the animals. The sentinel mouse cage is positioned closest to the air outlet. Healthy female mice(4-6 weeks old) are used for at least eight weeks in a cage with 100% dirty bedding. Monitoring sentinel mice quarterly includes bacterial, viral, parasitic, and necropsy testing.
In addition to standard microbiological surveillance, our staff observes the health of the animals daily, and any aberrant mice are referred to the examining veterinarian for clinical diagnosis, ensuring the mouse’s health and welfare.

Animal Welfare
The GemPharmatech Institutional Animal Care and Use Committee (IACUC) is responsible for supervising and evaluating all animal-related activities at GemPharmatech’s facilities to protect the animals’ welfare.
Our Surveillance System: Under the guidance of the Institutional Officer (IO), the GemPharmatech IACUC and the Attending Veterinarian (AV) supervise all animal and animal-related activities at the institution.
Our responsibilities: We supervise and offer regular training to staff on animal welfare and ensure the animals’ living conditions to enhance the welfare of all animals.
Our guidelines:
- Review all experimental protocols in the institution according to the 3R principles and provide approvals and oversight of the protocols.
- Supervise all animal-related activities in the institution.
- Provide animal welfare-related training to the institution’s personnel.
- Ensure a comprehensive animal welfare reporting mechanism is in place to detect and report any animal welfare violations in a timely manner.
Our daily work:
- Pre-approve experimental programs before commencement.
- Review experimental protocols following adoption.
- Regularly review animal activities and associated facilities within the institution.
- Conduct IACUC regular meetings at least once every two month to discuss animal welfare-related issues. Urgent meeting will be hold if necessary.
Standards:
Guide For The Care and use of Laboratory Animals Eight Edition
The IACUC HANDBOOK Second Edition
AVMA Guidelines for the Euthanasia of Animals: 2020 Edition
USDA Pain level
GBT 34791-2017 Laboratory animals-Requirement for quality control
GBT 35892-2018 Laboratory animals—Guideline for ethical review of animal welfare
Each mouse at GemPharmatech is unique in its life and worth, and we treat each mouse with the utmost respect and kindness.
For more information, please contact us [email protected]
Want To Inquire About The Services?
Contact Us

THE ATLANTIS BIOSCIENCE DIFFERENCE Discover Translational Solutions To Advance From Bench to Bed
GET SUPPORT Whenever You Need It

QUESTIONS IN YOUR MIND?
Connect With Our Technical Specialist.

KNOW WHAT YOU WANT?
Request For A Quotation.





