- Your cart is empty
- Continue Shopping
CryoEase-PF – A DMSO-Free, Protein-Free Cryopreservation Media for MSCs & Sensitive Cells
Dual-Temperature Compatible. Validated Performance. Translationally Ready.
CryoEase-PF is a next-generation, DMSO-free and protein-free cryopreservation medium designed specifically for the long-term preservation and short-term stability of human mesenchymal stem cells (MSCs) and other sensitive cell types.
What sets CryoEase-PF apart is its dual-temperature compatibility—the only cryomedia designed for both long-term storage in vapor-phase liquid nitrogen (-150°C) and short-term stability at 4°C for up to 72 hours. This breakthrough formulation enables real-world transport, decentralized trial execution, and cold chain simplification, ensuring your cells remain viable and functional — from cryobank to bedside.
Engineered in Singapore by AGEMBio, a cell therapy innovator, CryoEase-PF is optimized to meet the demands of biobanking, global cell therapy trials, and translational research workflows.
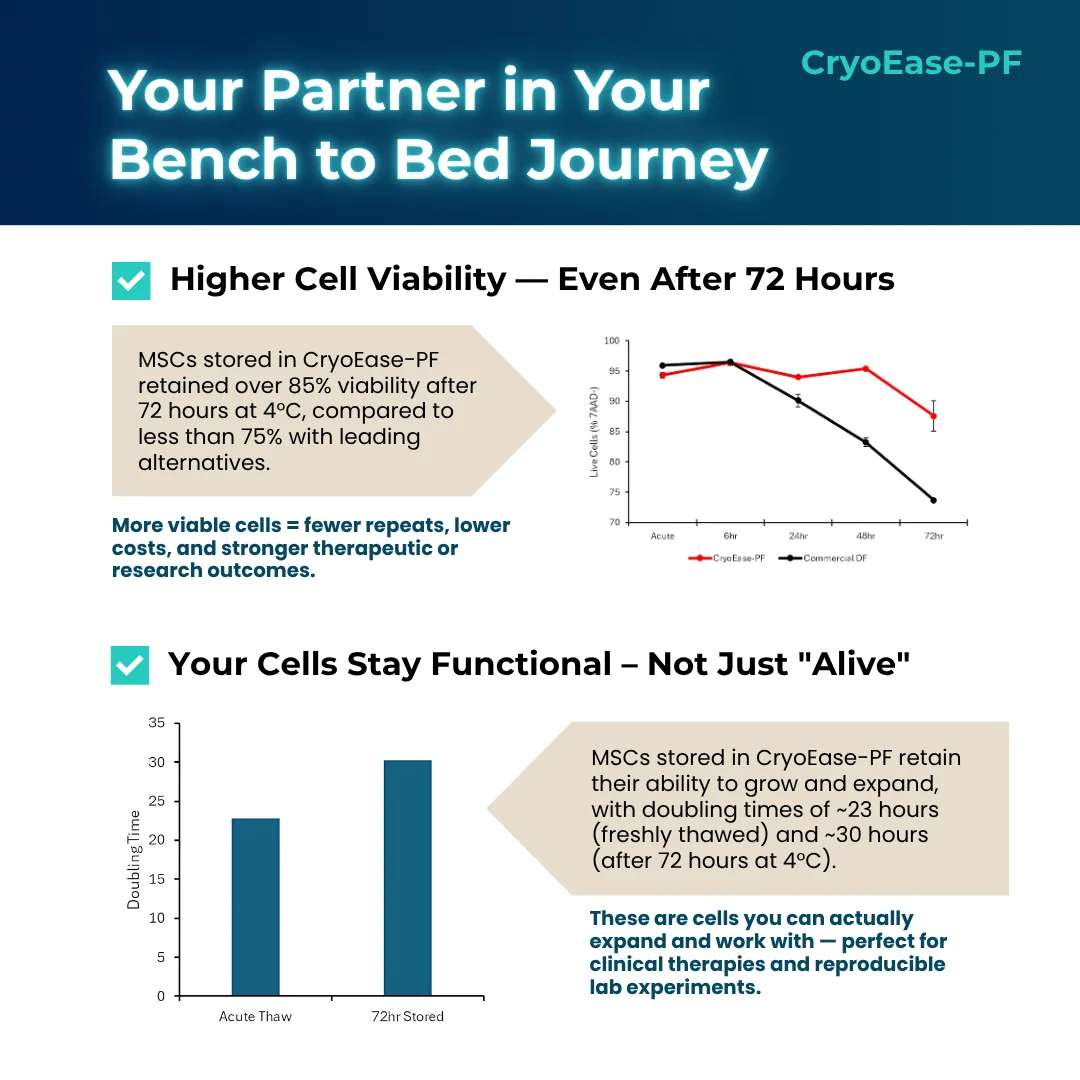
With >85% post-thaw viability and full ISCT-compliant phenotype retention even after 72 hours of hypothermic storage, CryoEase-PF provides unmatched protection for MSCs—from cryobank to bedside.
Validated Performance
- >85% Viability After 72 Hours at 4°C
Outperforms commercial cryomedia with <75% viability in the same conditions - 23-Hour Population Doubling Time Post-Thaw
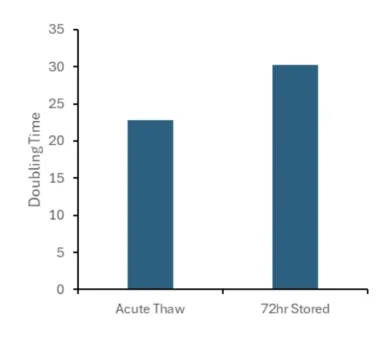
Comparable to fresh MSCs - 30-Hour PDT After 72h Hypothermic Hold
Ensures reliable cell expansion after transportation delays - ISCT-Compliant MSC Identity
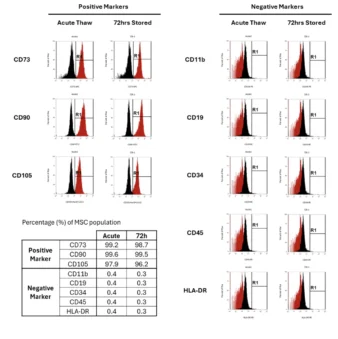
Retains CD73+/CD90+/CD105+ and CD34-/CD45-/HLA-DR- phenotype
Validation conducted using human adipose-derived MSCs (5 x 10⁶ cells/mL) stored at -150°C and held at 4°C for 72h post-thaw. Viability assessed via 7AAD staining.
 Dual-Temperature Compatibility: Real-World Ready
Dual-Temperature Compatibility: Real-World Ready
CryoEase-PF is uniquely optimized for translational logistics and multi-site trial workflows, allowing:
- Long-Term Biobanking: Vapor-phase LN₂ storage (-150°C)
- Short-Term Transport Stability: Maintains viability at 4°C for 72h
- Cold Chain Flexibility: Reduce reliance of dry ice and liquid nitrogen
- Allogeneic & Autologous Ready: Supports scalable, GMP workflows
This dual-temperature stability is unmatched in DMSO-free, protein-free cryopreservation — making CryoEase-PF the only truly translational-ready solution on the market.
Engineered for Translational Success
CryoEase-PF is formulated to support both allogeneic and autologous MSC workflows, ensuring functional and phenotypic consistency critical to IND filings, EMA submissions, and global trial logistics.
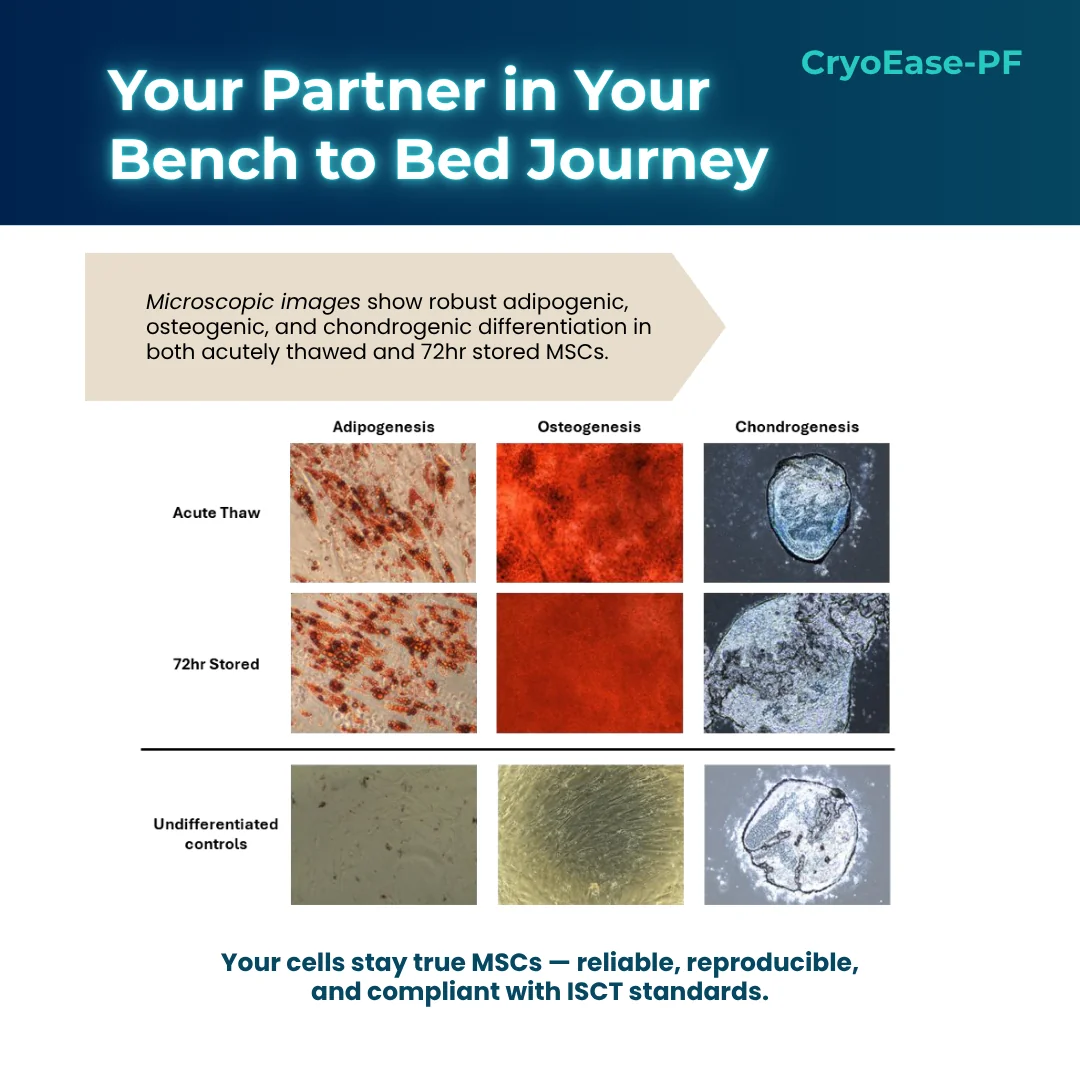
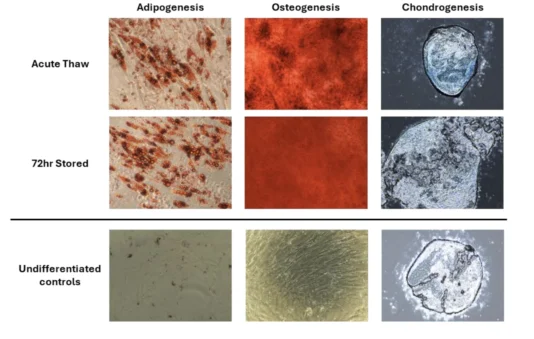
- Trilineage Differentiation Maintained – Osteogenic, chondrogenic, adipogenic
- Batch-to-Batch Consistency – Pre-mixed, endotoxin-tested
- No DMSO, No Proteins, No Risk – Eliminates toxicity concerns from cryoprotectants
Built for Global Deployment
Dual-temperature compatibility enables CryoEase-PF to simplify global logistics:
- Long-term: Storage in vapor-phase LN₂ (-150°C)
- Short-term: Viability at 4°C for up to 72 hours — ideal for multi-site advanced research studies and regional cell transport
CryoEase-PF vs. Conventional Cryomedia
| Parameter | CryoEase-PF | Typical Commercial Media |
|---|---|---|
| 72h Viability at 4°C | >85% | <75% |
| Post-Thaw PDT | 23h (acute), 30h (72h hold) | 35–50h (variable) |
| ISCT Compliance | ✅ Full retention | ❌ Often compromised |
| Cryoprotectant Risk | DMSO-free, xeno-free | DMSO-dependent |
Applications in MSC-Based Therapies
CryoEase-PF is engineered to meet the needs of every frontline advancing stem cell innovation:
- Clinical Settings:
Support MSC-based therapies with predictable and reproducible outcomes, critical for ensuring patient safety and therapeutic success. - Biobanking:
Preserve high-quality stem cells for long-term storage while maintaining viability and potency, ensuring reliable downstream applications. - Research Laboratories:
Enable robust, repeatable experiments by safeguarding stem cell integrity during cryopreservation and recovery. - Veterinary Medicine:
Facilitate advanced stem cell applications in animal health with DMSO-free, protein-free preservation optimized for sensitive cell types.
Developed by AGEMBio – Distributed by Atlantis Bioscience
CryoEase-PF is developed by AGEMBio, an NUS spin-off backed by SGInnovate, with a mission to redefine stem cell therapy through
immune-activating MSC platforms.
As exclusive distributor, Atlantis Bioscience supports the deployment of CryoEase-PF across APAC and beyond—aligning with our vision to deliver bench-to-bedside solutions that enable life-saving research, clinical translation, and scalable biomanufacturing.
Collaborate With Us – Expand CryoEase-PF to Your Cell Type
We’re inviting researchers and developers worldwide to explore CryoEase-PF’s capabilities across new cell types:
- ✓ Pilot studies with complimentary samples
- ✓ Collaborative method development
- ✓ Custom formulation support for sensitive cell lines
Contact us to join the validation network:
email: [email protected]
website: www.atlantisbioscience.com/contact-us/
| Form | Liquid |
|---|---|
| Brand | Atlantis Bioscience (Powered by AGEMBio) |
| Country of Origin | Singapore |
| Storage Condition | 4-8ºC |
| Quality Control | Certificate of analysis for each batch will be provided upon request |
| Volume | 100ml |
Supporting Data
To demonstrate the versatility of CryoEase-DF, we stored human adipose-derived MSCs at a density of 5 x 106 cells/mL. Upon thawing, the cells were stored at 4°C for up to 72 hours. Cell viability was assessed by 7AAD staining. Cells stored in CryoEase-PF retained >85% viability after 72 hours as compared to other commercially available formulations with <75% viability.

It is critical that the cells are not just viable but remain expandable in culture. MSCs frozen in CryoEase-PF were thawed and plated immediately (Acute Thaw) or stored at 4°C for up to 72 hours before being plated at 5,000 cells/cm2. The population doubling time (PDT) was determined after 4 days. Remarkably, cells stored in CryoEase-PF retain a PDT of approx. 23 hours and 30 hours when plated immediately or stored for 72 hours.
To ensure that MSC identity is not compromised post-storage, MSCs were subjected to phenotyping in accordance with the criteria set forth by the International Society for Cell and Gene Therapy (ISCT) (Dominici et. al.). MSC stored in CryoEase-PF retained both their surface marker profile and trilineage differentiation potential.
Protocol
Cryopreservation Protocol
- Trypsinize cells from a monolayer.
- Quantify cell number and viability.
- Pellet cells by centrifugation.
- Resuspend in an appropriate amount of CryoEase-PF.
- We recommend a freezing density of 1-5 x 10⁶ cells/mL
- Transfer to cryovials.
- Equilibrate at room temperature for 10-15 mins.
- Store at -80C.
- Optional: use control rate freezer/freezing devices for -1C/min freezing
Post-Thaw Sample Protocol
- Thaw vials in 37 °C water bath (or use automated thawing device – eg, ThawStar)
- Add complete media/appropriate thawing buffer at a 1:5 ratio (eg, 4 mL media to 1 mL frozen thawed cell suspension)
- Quantify cell number and viability.
- The sample is ready for downstream use. No spinning is required.
Documentation
Find supporting information and directions for use in the Product Information Booklet below.
MSC Workflow Bundle
Expand, Transport & Preserve
Atlantis Bioscience, in partnership with AGEMBio, offers a complete MSC solution that supports every step of your workflow — from high-efficiency cell expansion to safe short-term transport and long-term cryopreservation.
STEMGOLD – High-Performance hMSC Growth Medium Kit
- Accelerates MSC growth with faster doubling times
- Supports robust expansion of bone marrow, umbilical cord, and adipose-derived MSCs
- Preserves trilineage differentiation (osteoblast, chondrocyte, adipocyte)
- Eliminates pre-coating
- Ensure ISCT compliance
HypoGuard – The Global Standard for Hypothermic Cell Transport
- Preserves >95% MSC viability for 5 days at 2–8°C — no cryogenic shipping required
- Serum/Xeno & Protein-Free
- Suitable for sensitive cells (e.g. MSCs, PBMCs, etc.)
- Mysoplasma & Endotoxin Tested
- Ready to use – no special equipment or freezing required
Bundle These Products for Seamless MSC Research
From expansion (STEMGOLD) to transport (HypoGuard) to cryopreservation (CryoEase-PF), we provide a complete, integrated solution to support your translational MSC workflow — whether you’re running preclinical trials, global studies, or biobanking.
Ready to streamline your MSC process? Contact our team to discuss bundle pricing or sample requests.