- Your cart is empty
- Continue Shopping
Custom mRNA and Lipid Nanoparticles Services (RUO & GMP Available)
Messenger RNA (mRNA) is transforming modern medicine as a flexible and non-integrating platform for vaccines and gene therapies. Yet, its inherent instability requires advanced formulation and process control.
PackGene’s custom mRNA-LNP service, available in both RUO (Research Use Only) and GMP-compliant grades, integrates high-purity mRNA synthesis with optimized lipid nanoparticle (LNP) technology for enhanced stability, delivery, and safety.
Through our partnership, Atlantis Bioscience facilitates access to PackGene’s end-to-end solutions for researchers and biotech innovators across Singapore, Thailand, Malaysia, and Indonesia, ensuring seamless project support from discovery to clinical translation.
Fast turnaround
4-5 weeks from design to delivery
Custom Design
Tailored to your research needs
Flexible formats
RUO and GMP-grade production options available
Reduced immunogenicity
Minimised immune response for safer applications
Why Choose Our Custom mRNA-LNP Services?
With over 1,000 global clients and 50,000+ completed projects, PackGene delivers trusted excellence in nucleic acid manufacturing.
- 10+ years of expertise in viral and non-viral vector development
- 120,000 sq ft GMP facility, meeting FDA, EMA, and NMPA standards
- Proven performance and scalability from research to clinical production
Supported by Atlantis Bioscience as the regional partner in Southeast Asia, researchers can expect responsive communication, local consultation, and project management that bridges regional collaboration with PackGene’s global manufacturing capabilities.
How Our mRNA-LNP Is Made
Lipid nanoparticles (LNPs) are nanoscale lipid-based particles used as delivery vehicles for drugs, including mRNA. LNP-based methods are the most popular for mRNA delivery, offering benefits such as enhanced stability, improved delivery efficiency, reduced immunogenicity, customizability, and a proven clinical success record. These advantages make LNPs a promising tool for the development of safe and effective mRNA-based treatments and vaccines.
PackGene’s workflow is designed to ensure efficiency, reproducibility, and full traceability for regulatory submission.
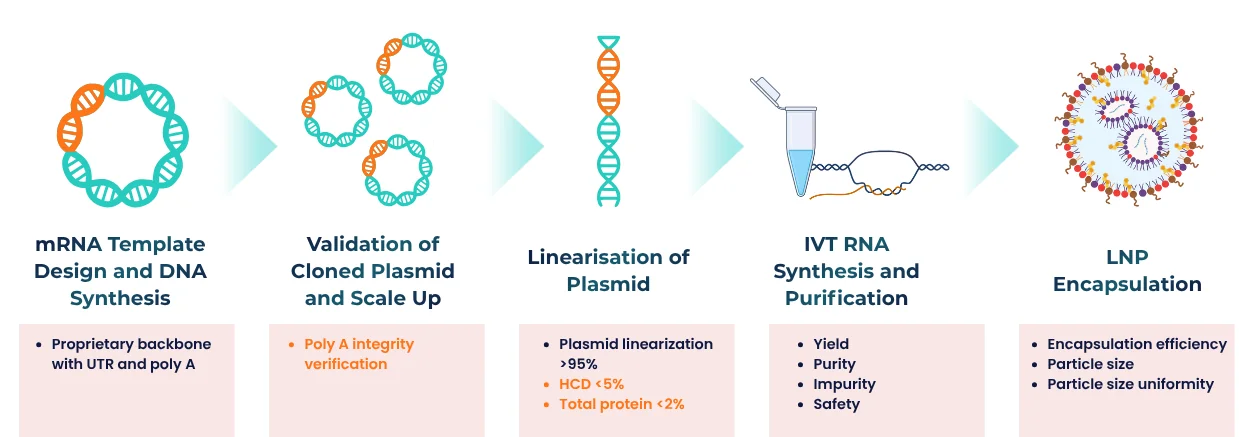
Custom mRNA production using in vitro transcription (IVT) is a proven method for synthesizing high-quality RUO mRNA and GMP mRNA from plasmid DNA templates. PackGene’s IVT process incorporates a promoter sequence, the gene of interest, and a poly(A) tail, streamlining mRNA manufacturing while ensuring consistency, scalability, and cost-effectiveness for research, preclinical, and clinical applications.
What We Offer
| Catalog | Capping and modification | Gene | Quantity | Timeline (Business days) |
|---|---|---|---|---|
| mRNA | Custom mRNA Cap1 | Custom gene | 100μg-20mg | 10-15 |
| mRNA | Custom mRNA Cap1 N1meΨU | Custom gene | 100μg-20mg | 10-15 |
| mRNA-LNP | Custom mRNA Cap1 in LNP | Custom gene or off-the-shelf | 100μg-20mg | 10-25 |
| mRNA-LNP | Custom mRNA Cap1 N1meΨU in LNP | Custom gene or off-the-shelf | 100μg-20mg | 10-25 |
*Additional time (~2-3 weeks ) for custom gene synthesis
mRNA grade and QC standard
| QC Category | QC Item | Method | Specification | Research Grade |
|---|---|---|---|---|
| mRNA-LNP (On-shelf & Custom) | RNA Integrity / Size | Capillary Electrophoresis | > 80 % | ✓ |
| Encapsulation Efficiency | ELISA Analyzer | Report | ✓ | |
| Particle Size | Dynamic Light Scattering | Z Ave. Dia < 120 nm | ✓ | |
| Particle Uniformity | Dynamic Light Scattering | PDI < 0.2 | ✓ | |
| Concentration | Capillary Electrophoresis | Encapsulated mRNA > 0.2 mg/mL | ✓ |
PackGene’s quality control (QC) testing of mRNA-LNPs includes measurement of various parameters to ensure their quality, safety, and efficacy. Some of the essential QC tests for mRNA LNP are encapsulation efficiency, particle size, and uniformity:
Encapsulation Efficiency (EE)
EE measures the percentage of mRNA encapsulated within LNPs, essential for effective delivery.
Encapsulation Efficiency (EE) = (Amount of encapsulated mRNA / Total amount of mRNA) x 100%
For example, if 1 mg of mRNA yields 0.8 mg encapsulated, EE = 80%. High EE ensures optimal therapeutic outcomes.
Particle Size
Particle size impacts biodistribution, cellular uptake, and stability. Larger particles may aggregate, increasing toxicity. Dynamic light scattering (DLS) is commonly used to measure size.
Size Distribution and Uniformity
A narrow size distribution enhances stability and consistency, while variability may lead to toxicity. Methods like DLS and nanoparticle tracking analysis (NTA) assess these parameters.
These tests are critical for ensuring safe, effective LNP-based mRNA delivery.
Applications of LNPs
Increased Stability
Enhanced Delivery
Reduced Immunogenicity
Customization
Clinical Success
LNPs protect mRNA from degradation by enzymes and other factors in the body, enhancing its stability and improving delivery to target cells.
LNPs enhance the efficient uptake of mRNA by cells, resulting in higher protein expression levels. They also enable targeted delivery of mRNA to specific cells, such as immune or tumor cells, through cell-type specific endocytosis mechanisms.
LNPs can lower the immunogenicity of mRNA by protecting it from the immune system and preventing the activation of innate immune responses. This enhances the safety and efficacy of mRNA-based therapies and vaccines.
The composition of LNPs can be tailored to enhance properties like stability, delivery efficiency, and immunogenicity. This enables customization for specific applications, such as vaccines or therapies aimed at particular diseases.
LNPs have been used successfully in the development of mRNA vaccines for COVID-19, showing high efficacy and safety in clinical trials. This success has increased interest in the use of LNPs for other mRNA-based therapies and vaccines.
Case Study
EGFP mRNA (Cap1) -LNP
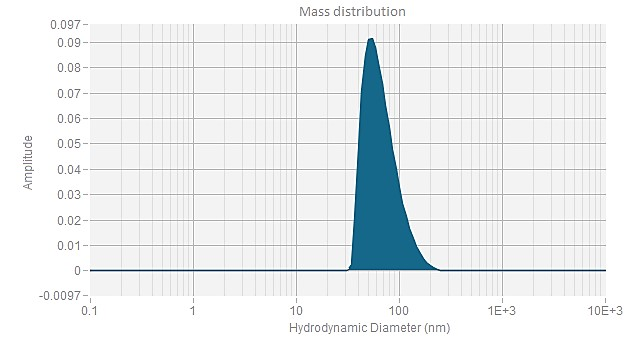
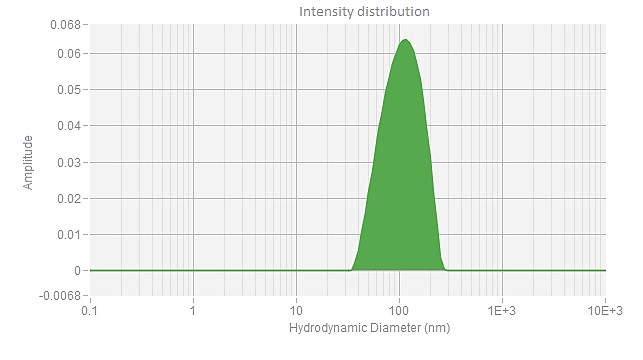
| Z Ave. Dia (nm) | 76.8 |
|---|---|
| PDI | PDI |
| Encapsulation Efficiency | 93.91% |
The Z-average diameter of nanoparticles is determined using dynamic light scattering (DLS), which measures the Brownian motion of particles in solution. By analyzing light scattering fluctuations, the size distribution of the particles is calculated. The Z-average diameter is the intensity-weighted mean size, with larger particles contributing more to the value. This parameter, typically reported in nanometers (nm), reflects the overall size of the particle population. PackGene guarantees an mRNA-LNP Z-average diameter of <120 nm and an encapsulation efficiency of >90%.
The polydispersity index (PDI) measures the size uniformity of nanoparticles. A low PDI value indicates a narrow size distribution, while a high PDI suggests greater size variability. In mRNA delivery, a low PDI is desirable for improved stability, efficacy, and safety. PackGene guarantees a PDI of <0.2 for its mRNA-LNP products, ensuring consistency and high quality.
Injection of mRNA-LNP to animals
Firefly luciferase mRNA9(Cap1)-LNP
Live image 6hrs after mouse intravenous injection

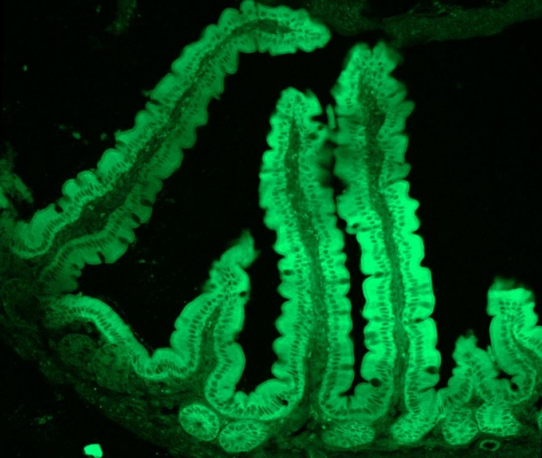
EGFP mRNA9(Cap1)-LNP
Confocal image of mouse intestine frozen section 24hrs after enema
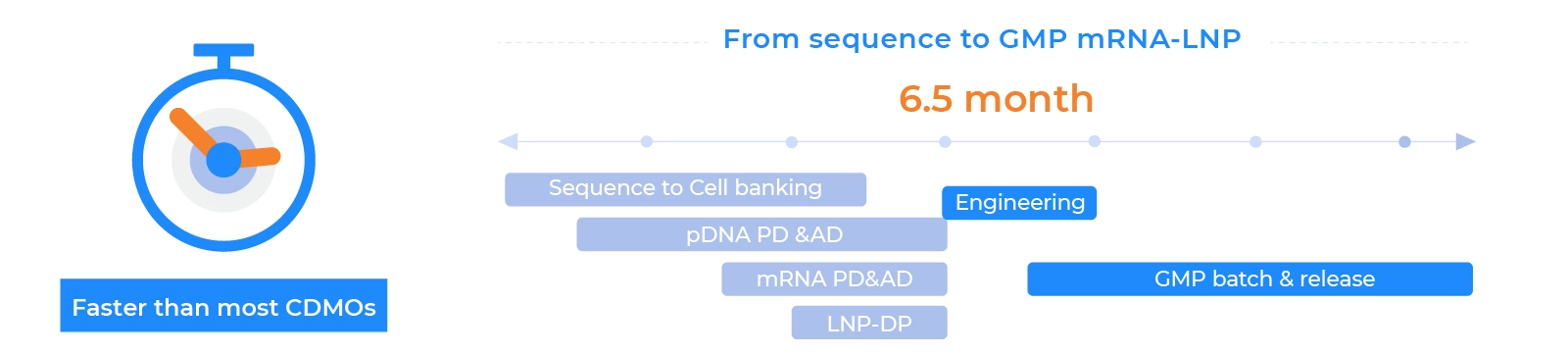
GMP mRNA Manufacturing — From Development to Clinical Readiness
PackGene offers a comprehensive GMP mRNA & LNP manufacturing platform, supporting your journey from early development to clinical production. With advanced facilities and a robust quality management system, we ensure every batch meets the highest standards of purity, consistency, and regulatory compliance.
Key Features
End-to-end capability
From plasmid construction and mRNA & LNP synthesis to formulation development, fill and finish, and full documentation.
Seamless scale-up
Smooth transition from Process Development (PD) to cGMP manufacturing for research and clinical phases.
Customised process optimisation
Tailored workflows to maximise mRNA & LNP yield, stability, and reproducibility.
Global regulatory compliance
Operates under a QMS aligned with FDA, EMA, and NMPA standards.
Comprehensive analytical support
Includes in-process control, release testing, and stability studies for full traceability.
Regulatory-ready quality
Delivering GMP-grade mRNA & LNP materials suitable for IND filing and preclinical studies.
What We Offer
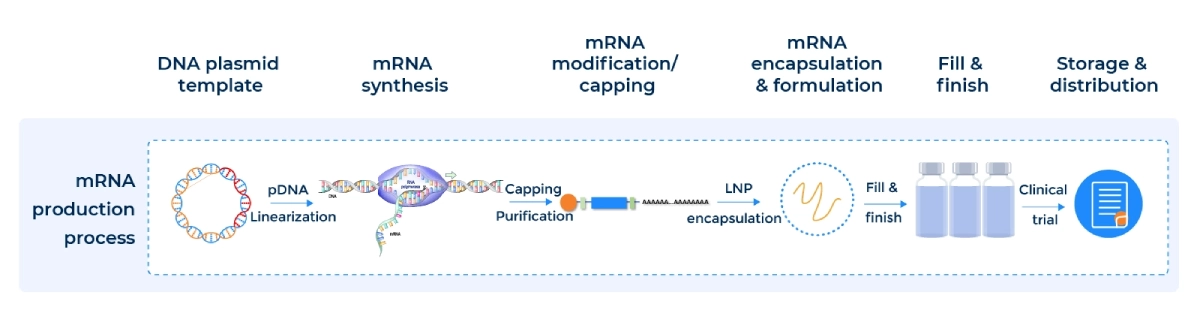
For all your mRNA manufacturing needs. Our streamlined process ensures ease in your CMC
Product Quality and Speed


Comprehensive Analytical Panel
Packgene’s comprehensive analytical panel allows for customized in-process control, release testing, and stability studies for plasmid, mRNA, and LNP. This enables us to tailor our approach to meet the specific needs of your project and ensure that the final product is of the highest quality.
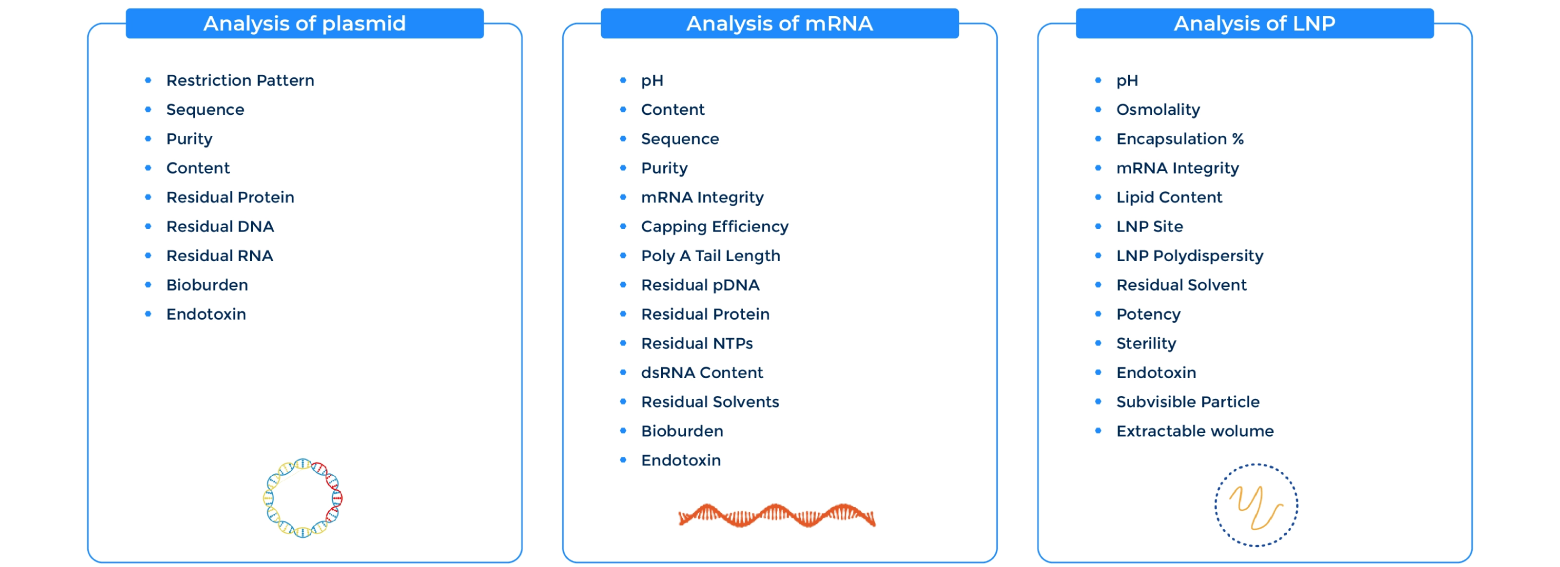
With our advanced analytical tools, Packgene monitor every step of the production process to identify any potential issues early. This allows us to take corrective action quickly and efficiently when necessary. Packgene commitment to using the latest analytical techniques means that you can have confidence in the accuracy and reliability of our testing methods.
GMP Documentation & Traceability
PackGene maintains complete documentation to ensure full regulatory compliance and consistent product quality. Every stage of mRNA & LNP manufacturing is traceable through well-structured project, operation, and quality records — supporting smooth transitions from R&D to clinical production.
| Project Document | Operation Document | Quality Document |
|---|---|---|
| Enquiry Assessment | SOPs | Specification |
| Manufacture Summary | Goods Receiving Record | Material and Product Test |
| Packaging Summary | Work Order | CoA |
| Delivery Summary | Batch Record | Batch Record Review and Release |
| Agreement and Contract | Shipping Request | Stability Data |
| Distribution Record | Validation Report | |
| Logbook | Supplier Qualification | |
| Environment Monitor |
Frequently Asked Questions
Can PackGene co-encapsulate both gene-editing mRNA and sgRNA in LNP? Which LNP formulation should be used?
Yes, we can co-encapsulate gene-editing mRNA and sgRNA in LNP. We have successfully co-encapsulated SpCas9 mRNA with sgRNA, achieving robust gene-editing efficiency in Huh7 cells. We recommend using the LP01 LNP formulation, and we will provide updates as we test other formulations.
How much mRNA in LNP is needed for mouse injections?
The amount of mRNA depends on the desired expression level and target cells or tissues. For mouse injections, we have tested 0.5 mg of FLuc or EGFP mRNA in LNP per kg of mouse body weight, yielding strong expression. For specific genes, it’s best to try a few different doses to optimize the amount.
What is the concentration of mRNA in LNP?
Our standard mRNA concentration in LNP is 0.2 mg/mL, but we can increase this to 2 mg/mL. If higher concentrations are needed, please contact our technical support team.
Q: How should I store and handle mRNA in LNP?
mRNA-LNP can be stored at -80°C for up to 2 years. However, mRNA-LNP is highly sensitive to freeze-thaw cycles, which can compromise its ability to transduce cells or tissues. To avoid this, please inform us of your preferred aliquot sizes. Our pre-aliquoted mRNA-LNP kits come in volumes of 10 µL or 50 µL.
Trusted mRNA-LNP Partner in Southeast Asia
PackGene’s proven mRNA-LNP manufacturing expertise is now locally accessible through Atlantis Bioscience, your regional bridge to world-class nucleic acid production. Whether you are exploring early-stage mRNA research or preparing for GMP-grade translation, our partnership ensures faster communication, regional support, and streamlined logistics — from Singapore to Thailand, Malaysia, and Indonesia.
Together with PackGene, we are committed to breaking barriers in gene therapy by providing comprehensive and cost-effective AAV, lentivirus, mRNA and plasmid solutions.
Contact Us Today to accelerate your next breakthrough with a reliable CDMO partner who understands your workflow.
Want To Inquire About The Services?
Contact Us

THE ATLANTIS BIOSCIENCE DIFFERENCE Discover Translational Solutions To Advance From Bench to Bed
GET SUPPORT Whenever You Need It

QUESTIONS IN YOUR MIND?
Connect With Our Technical Specialist.

KNOW WHAT YOU WANT?
Request For A Quotation.





